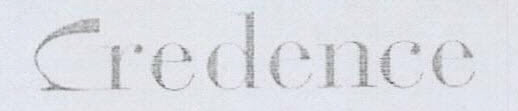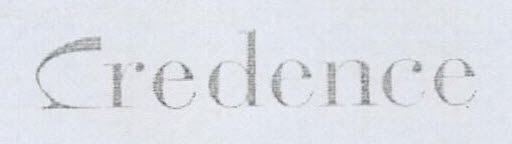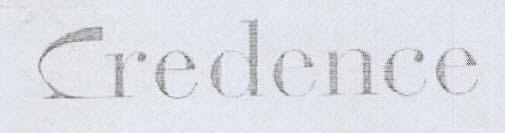Company Information
CIN
Status
Date of Incorporation
15 June 1989
State / ROC
/ ROC Hyderabad
Industry
Sub Category
Last Balance Sheet
Last Annual Meeting
Paid Up Capital
610,305,680
Authorised Capital
1,000,000,000
Directors
Past Directors
Patents
A simple and scaleable process is provided for obtaining recombinant human GM-CSF expressed in microbial cells, secreting mature GM-CSF, culturing these cells and purifying the protein in good yield and of a therapeutic grade quality. The steps include cloning, fermentation and purification of the protein. The purif...
Humanized monoclonal antibodies against human epidermal growth factor receptor, use and method thereof
Humanized MAbs are designed, created and produced using a series of innovative processes comprising of computational analysis, modeling studies, rationale assessment and selection of humanized sequences, an effici...
The present invention discloses a process for stable, liquid therapeutic formulation of G-CSF, wherein the protein is stored in a buffer of conductivity greater than 1.0 milliSiemens/cm and in the pH range of 4.0 to 5.0, in the presence of a surfactant, a tonicity modifier and an amino acid that suppresses the oxida...
The present invention provides a method for achieving high-level expression of the therapeutically important Iymphokine (human IL-2). The method comprises of identifying the secondary structure in the 5' region of human IL-2 mRNA, modifying the 5' region of the human IL-2 DNA sequence to produce a new DNA sequence w...
The present invention describes a novel process for large scale purification of therapeutic grade quality of recombinant human G-CSF from microbial cells, wherein the protein is expressed as inclusion bodies. The process involves the novel use of Hydrophobic Interaction Chromatography (HIC) step to purify G-CSF elut...
Trademarks
[Class : 5] Pharmaceutical And Medicinal Preparations For Human And Veterinary Use
[Class : 5] Pharmaceutical And Bio Pharmaceutical Preparations.
["Trade mark is likely to be removed due to non filing of Renewal request within prescribed time limit In case of any discripancy contact respective TM Registry. "]
[Class : 5] Pharmaceutical And Bio Pharmaceutical Preparations All Being Goods Included In Class 5.
[Class : 5] Pharmaceutical And Bio Pharmaceutical Preparations.
[Class : 5] Pharmaceutical And Medicinal Preparations For Human And Veterinary Use
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed Matter Plastic And Other Material For Packaging All Being Goods Included In Class 16
[Class : 5] Pharmaceutical And Bio Pharmaceutical Preparations.
[Class : 5] Medicinal And Pharmaceutical Preparations And Substances And All Being Goods In Class 5
[Class : 5] Pharmaceutical And Bio Pharmaceutical Preparations Included In Class 5.
[Class : 5] Pharmaceutical And Bio Pharmaceutical Preparations.
[Class : 5] Medicinal And Pharmaceutical Preparations And Substances And All Being Goods In Class 5
[Class : 42] Pharmaceutical Research, Toxicity Studies And Bio Equivalence Studies, Clinical Trials, Clinical Researches, Clinical Testing Of The Experimental Drugs, Clinical Laboratory And Bio Analytical Services Being Services Included In Class 42.
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed Matter Plastic And Other Material For Packaging All Being Goods Included In Class 16 (Sixteen)
["Trade mark is likely to be removed due to non filing of Renewal request within prescribed time limit In case of any discripancy contact respective TM Registry. "]
[Class : 5] Pharmaceutical And Medicinal Preparations For Human And Veterinary Use.
[Class : 5] Pharmaceutical And Bio Pharmaceutical Preparations
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 16] Cardboard And Goods Made From Other Material, Printed, Plastic And Other Material For Packaging,
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 42] Pharmaceutical Research, Toxicity Studies And Bio Equivalence Studies, Clinical Trials, Clinical Researches, Clinical Testing Of The Experimental Drugs, Clinical Laboratory And Bio Analytical Services.
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed, Plastic And Other Material For Packaging,
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed, Plastic And Other Material For Packaging
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 5] Pharmaceutical And Bio Pharmaceutical Preparations
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed, Plastic And Other Material For Packaging
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed, Plastic And Other Material For Packaging
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed, Plastic And Other Material For Packaging
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 5] Pharmaceutical And Bio Pharmaceutical Preparations
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed, Plastic And Other Material For Packaging
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed, Plastic And Other Material For Packaging
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed, Plastic And Other Material For Packaging
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 16] Paper, Cardboard And Goods Made From Other Material, Printed, Plastic And Other Material For Packaging
["Renewal of trademark is due. Renewal request in prescribed form is to be filed by the applicant. "]
[Class : 5] Pharmaceutical And Bio Pharmaceutical Preparations Included In Class 05.